Nuclear Medicinal Chemistry & Radiometal Chemistry
|
Nuclear medicine is a powerful technique with the ability to image disease non-invasively and subsequently treat the diseased tissue by injecting a radioactive isotope fused to a disease-targeting biomolecule. The targeting vector (aka biomolecule) is often a small molecule, peptide, or antibody which exhibits high affinity for over-expressed surface receptors on diseased cells (e.g., cancer cells). Covalent attachment of the radionuclide to a biomolecule ensures that the radioactive payload is specifically and efficiently delivered to the cells or tissue that a physician needs to image or treat. Our lab exploits metallic radionuclides (aka radiometals) and their diverse radioactive decay properties for both imaging and therapy of disease. For example, radioisotopes that decay via gamma ray or positron emission can be used for single-photon emission computed tomography (SPECT), or positron emission tomography (PET) imaging, respectively. Radioisotopes that emit beta particles, alpha particles, or Meitner-Auger electrons can be used for targeted radionuclide therapy (TRT), since these radioactive emissions are associated with high linear energy transfer (LET), and can cause harm and kill target cells.
Current research projects include 1) medical isotope production, 2) radiometal chelation, and 3) radiopharmaceutical development. |
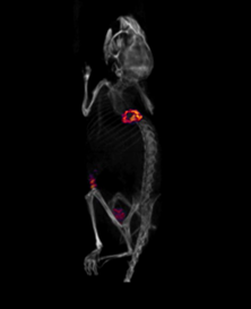
SPECT/CT image of tumour bearing-mouse injected with In-111 labeled antibody (Trastuzumab) taken 3 days post-injection
|
1) Medical Isotope Production
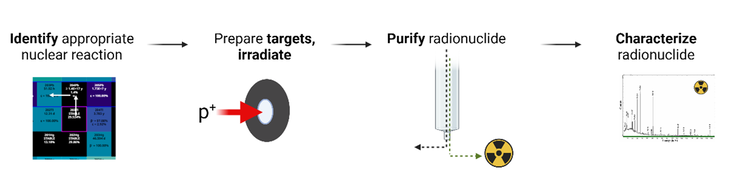
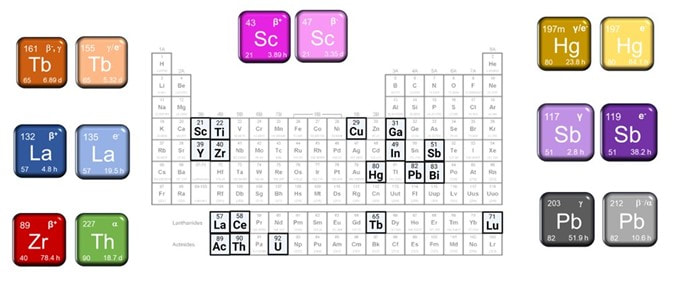
Enabled by unique particle accelerator infrastructure at TRIUMF, we produce, isolate, and characterize exotic radiometals that have promising physical decay properties for incorporation into a radiopharmaceutical for imaging or therapy. This research entails (1) targetry - preparing targets from stable (non-radioactive) starting materials that can withstand the extreme conditions encountered during proton irradiation, (2) radiochemical separation – developing methodology that can chemically separate the mg-g amounts of target material from the ng-ug amount of radionuclide produced in a form suitable for subsequent radiolabeling and radiopharmaceutical development, and (3) characterizing the isolated radiometals for radionuclidic and chemical purity using a variety of (radio)analytical techniques. These projects are often in collaboration with TRIUMF Scientists.
Examples of Research
2) Radiometal Chelation
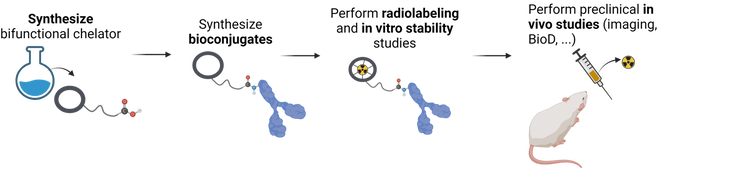
Many of the radiometal candidates with promising physical decay properties also have limited or unexplored chelate chemistry with respect to their radiolabeling properties. A critical component of a metal-based radiopharmaceutical is the bifunctional chelator – used to bind the radiometal in a stable and inert coordination complex.
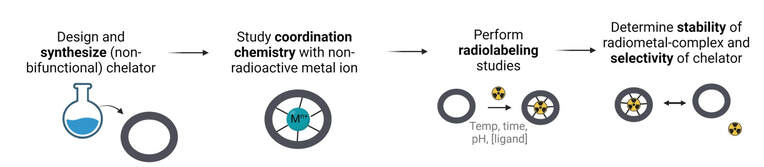
We use our expertise in inorganic chemistry and coordination chemistry to design, synthesize, and evaluate novel chelating ligands and their radiometal ion complexes in vitro, ex vivo, and in vivo.
We investigate the non-radioactive (stable) metal-complex using conventional techniques (NMR, MS, X-ray, potentiometry) to gain fundamental insight into the binding environment. We test our chelators with the myriad of radiometal ions produced and available to us, and perform in depth radiolabeling and in vitro stability assays.
We use our expertise in inorganic chemistry and coordination chemistry to design, synthesize, and evaluate novel chelating ligands and their radiometal ion complexes in vitro, ex vivo, and in vivo.
We investigate the non-radioactive (stable) metal-complex using conventional techniques (NMR, MS, X-ray, potentiometry) to gain fundamental insight into the binding environment. We test our chelators with the myriad of radiometal ions produced and available to us, and perform in depth radiolabeling and in vitro stability assays.
Examples of Research
3) Radiopharmaceutical Development
As an ultimate test of our chelators as carriers of radiometals in radiopharmaceutical design, we perform targeted studies with prepared bioconjugates. Bifunctional chelators are synthesized and conjugated to targeting vectors (e.g., small molecules, peptides, antibodies) that have high affinity to over expressed surface receptors on cancer cells. We carefully select and test appropriate linkers and bioconjugation strategies for the covalent attachment of the bifunctional chelator to the targeting vector. In vitro (cell binding, internalization), and in vivo (preclinical imaging, biodistribution, and therapy) studies are conducted.